Lithium-ion batteries

What are lithium-ion batteries?
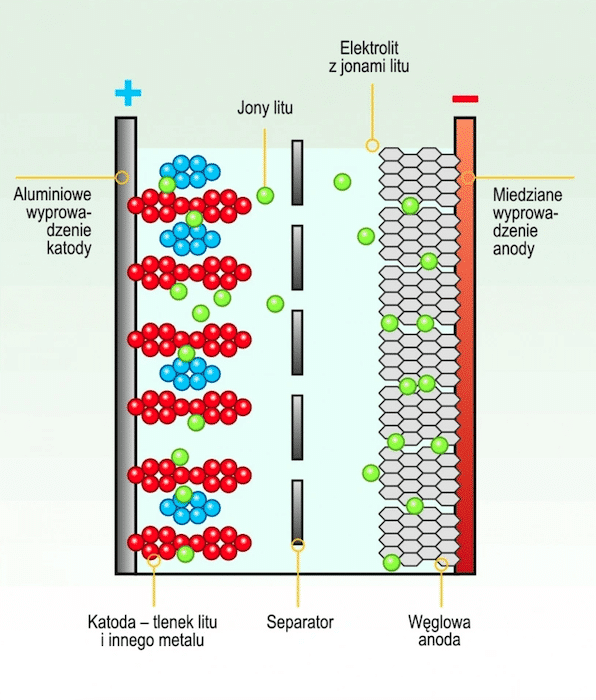
A lithium-ion battery (often referred to as a lithium-ion or Li-ion battery) is one of the most widely used types of rechargeable batteries today. Its name is derived from its fundamental operating principle: during discharge, lithium ions migrate from the negative electrode (anode) through the electrolyte to the positive electrode (cathode) while electrons flow through the external circuit. When the battery is being charged, the direction of ion movement is reversed.
Lithium-ion battery operation. Source: Computerworld
Characteristic features and quantities
Lithium-ion batteries stand out from other secondary cells due to their high gravimetric and volumetric energy density, which means they can store a significant amount of energy per unit of mass or volume. This exceptional property is a result of their use of non-aqueous electrolytes, which can be in the form of liquid solutions containing lithium salts mixed with organic solvents or solid materials. This characteristic makes lithium-ion batteries suitable for use in small, lightweight mobile electronic devices and electric vehicles.
In addition to their high energy density, lithium-ion batteries offer other advantages, including the absence of a memory effect. The memory effect is the real or apparent loss of capacity that can occur in some batteries due to incomplete discharge or excessive charging. However, it’s worth noting that this effect is not present in lithium iron phosphate (LFP) cells.
Some of the quantities that define lithium-ion batteries are:
– stored energy: most often expressed in watt-hours (Wh) and related units, also called capacity,
– capacity: the amount of charge that can be stored in the battery, expressed in ampere-hours (Ah) and derived units,
– efficiency: the ratio of the amount of energy that can be obtained from the battery to the energy supplied during charging, expressed as a percentage,
– service life: the number of charge and discharge cycles after which the battery capacity drops below a certain level,
– charge status: (SoC – state of charge) expressed as a percentage, the ratio of the current battery charge level to its total capacity,
– discharge depth: (“DoD” depth of discharge) battery discharge level expressed as a percentage; it is important not to exceed the value recommended by the manufacturer so as not to shorten the battery life,
– wear condition: (SoH – state of health) the ratio of the maximum battery charge to the nominal capacity expressed as a percentage; decreases with the number of charge and discharge cycles,
– recommended charging/discharging current: the range of current at which the battery should be charged/discharged to avoid damage or premature deterioration of its parameters; expressed as a fraction of the nominal capacity (C), where 1C is the so-called one-hour current, i.e. the current at which the discharged battery would operate for one hour.
The history of lithium-ion batteries
In 1958, WS Harris conducted research on the solubility of lithium in non-aqueous electrolytes. During his study, he noticed the development of a passive layer. This layer acted as a barrier, preventing a direct interaction between lithium and the electrolyte while still enabling the passage of ions. This discovery sparked greater enthusiasm for the potential application of lithium-ion batteries.
During the early 1970s, there was significant research into the intercalation reaction, which involves the reversible penetration of ions, atoms, or molecules from another substance into the crystal structure. This research also explored the concepts of energy storage in secondary cells based on lithium. In the subsequent decade, lithium batteries employing titanium disulfide as a cathode were developed and brought to the commercial market. However, these batteries had a drawback: they produced low voltage, around 2.5 volts, resulting in limited energy density.
As a response to this limitation, scientists began searching for alternative cathode materials. This quest led to the identification of three categories of oxide cathodes. Among these categories, layered oxides were found to possess the highest energy density. Today, they are commonly used, with LiCoO2 being one of the most frequently employed materials.
Initially, lithium and its alloys were employed as the anode material. However, this approach had a drawback, as it could lead to the formation of dendrites—crystals that grow in a single direction and could damage the separator between the electrodes. Consequently, there was a consideration for using two distinct materials for the production of the anode and cathode.
This idea was put into practice in 1991 when a lithium-ion battery featuring a graphite anode was introduced for commercial production. To this day, it remains the most prevalent type of anode used in lithium-ion batteries.
Since the commercialization of lithium-ion battery applications, it has been continually improved to better meet consumer demands.
Types of lithium-ion batteries
There are several types of lithium-ion batteries chosen for specific applications based on factors such as power needs, efficiency, and operating conditions. These types include:
- LCO (Lithium-Cobalt Oxide): These batteries are known for their high energy density and safety but have limitations in charging and discharging current.
- LFP (Lithium Iron Phosphate): LFP batteries have a low voltage but can handle high current, making them suitable for applications requiring a quick power surge.
- LMO (Lithium Manganese Oxide): These batteries offer high charging and discharging currents but come with lower energy density and service life.
- NCA (Lithium-Nickel-Cobalt-Aluminum): NCA batteries provide both high energy density and power, along with a longer lifespan. However, they tend to be costlier and have lower safety levels compared to other lithium-ion types.
- NMC (Lithium-Nickel-Manganese-Cobalt): NMC batteries are versatile and can be optimized for either high energy density or power, depending on the specific application requirements.
Use of lithium-ion batteries
Portable electronic devices
Lithium-ion batteries, owing to their exceptional energy and power density, serve as the ideal power source for a wide range of small electronic devices like mobile phones, laptops, and tablets. Furthermore, they are a prevalent choice for numerous medical devices, including hearing aids, infusion pumps, pacemakers, and defibrillators.
UPS systems
Lithium-ion batteries play a crucial role in providing uninterrupted power in UPS systems (Uninterrupted Power Systems). These systems are essential for devices critical to human life, such as medical equipment in hospitals. Additionally, they are widely employed to safeguard against potential disruptions like data loss on IT servers and the loss of unsaved work progress in office settings.
Electric vehicles
The characteristics of lithium-ion batteries also position them as an excellent choice for powering vehicles like cars, bicycles, and electric scooters. These modes of transportation are gaining popularity due to rising liquid fuel costs, a commitment to environmental conservation by users, and ongoing advancements in fast and convenient charging technologies. Furthermore, battery-powered vessels such as electric boats and yachts are seeing a surge in popularity as well.
Energy storage
Lithium-ion batteries play a fundamental role in energy storage to manage grid load balancing. They are especially indispensable in renewable energy systems, where energy storage is frequently required at a smaller scale, such as in conjunction with residential solar panels. Their utilization enables the efficient harnessing of energy from uncontrollable sources by storing excess energy when it’s generated and returning it to the grid when there’s a demand for it. This helps optimize the utilization of renewable energy resources.
Hazards related to the use of lithium-ion batteries
Explosion hazard
The electrolytes used in lithium-ion batteries are indeed flammable, posing a risk of explosion if their structure is compromised or if they’re subjected to excessive current during charging. Additionally, lithium-ion batteries have a specific voltage operating range, typically between 2.5 to 3.65 volts (and up to 4.35 volts in some cases). Charging these batteries improperly can lead to voltage exceeding the safe limits, potentially reducing battery capacity and, in extreme cases, causing a dangerous explosion due to reactive components.
Furthermore, storing lithium-ion batteries for extended periods without use can result in premature capacity loss. This can create a risk of overcharging during subsequent use, even if the user follows the manufacturer’s charging instructions. Proper handling, charging, and storage are essential to mitigate these safety concerns associated with lithium-ion batteries.
Indeed, safety standards for the manufacturing and testing of lithium-ion batteries are exceptionally rigorous. Before lithium-ion batteries can be transported and used, they must undergo a battery of tests. These tests encompass a wide range of scenarios, including height simulation, thermal tests, crushing, vibration, impacts, internal short circuit tests, overload simulations, and forced discharge assessments. These stringent safety measures are in place to ensure the safe production and handling of lithium-ion batteries, minimizing the risk of accidents and ensuring their reliability in various applications.
Thermal runaway and spontaneous combustion
When a lithium-ion battery overheats to an extreme extent, it can trigger a phenomenon known as thermal runaway. This is an uncontrolled chain reaction that begins with the evaporation of the battery’s electrolyte, leading to a surge in pressure within the battery. Consequently, flammable gases can escape from the battery, and these gases can ignite upon contact with air. When multiple cells are placed in proximity to one another, a rise in temperature in one cell can be sufficient to propagate the chain reaction to adjacent cells. Managing a battery fire is extremely challenging, often necessitating the containment of the fire’s spread until the flammable substances are fully exhausted.
Battery fire hazards can arise from manufacturing defects, improper storage, or mishandling of batteries. This underscores the critical importance of rigorous testing of batteries before they are approved for use, as well as the need to protect batteries, especially against electrical and thermal overloads, and mechanical damage during operation. Safety measures and protocols are crucial to prevent these potential hazards associated with lithium-ion batteries.
Lithium-ion batteries and the environment
Extraction of raw materials for the production of lithium-ion batteries
Lithium-ion batteries are often associated with environmentally friendly applications, but their production does indeed raise ecological concerns. These batteries rely on component materials like cobalt and lithium, which are considered rare metals. Unfortunately, the extraction of these metals, often in developing countries, can have significant environmental impacts. Mining technologies used in these regions can lead to environmental degradation and pose ecological challenges.
For example, a substantial portion of the world’s lithium resources is concentrated in the Andes region of Argentina, Bolivia, and Chile. The extraction process for lithium is water-intensive, requiring nearly 2 million liters of water for the production of one tonne of lithium. This can lead to the depletion of local groundwater resources, soil pollution, and other forms of environmental damage. As a result, many local residents, who primarily rely on agriculture, are often forced to leave their homes. Furthermore, global demand for lithium is expected to rise, intensifying these issues.
Cobalt, another essential component of lithium-ion batteries, is extracted significantly in the Democratic Republic of the Congo. Unfortunately, local mining practices do not adequately protect against the contamination of rivers and water bodies with sulfur compounds. When these compounds come into contact with water, they can form sulfuric acid, posing a danger to local fauna and human populations. Additionally, cobalt mining may release dust containing toxic metals, including uranium.
Graphite mining, associated with battery production, can also contribute to environmental concerns. The use of explosives in the mining process can lead to the dispersion of harmful substances, resulting in local air and soil pollution.
Efforts are underway to develop more sustainable and environmentally friendly practices in the extraction and production of these materials for lithium-ion batteries. These initiatives aim to reduce the ecological footprint associated with battery manufacturing.
Recycling of lithium-ion batteries
Due to the hazards linked to improper storage and disposal of lithium-ion batteries, it’s imperative that they are never discarded in landfills or treated as regular waste, such as plastic. Instead, these batteries should be directed to certified companies that specialize in their safe disposal and material recovery processes. According to Fortum, it is feasible to reclaim as much as 95% of valuable materials like lithium, cobalt, and graphite from a used battery. In total, about 80% of the lithium-ion battery’s components can be recycled, making responsible disposal and recycling crucial for minimizing environmental impact and maximizing resource recovery.
Alternatives to lithium-ion batteries
Indeed, ongoing research and development are continually exploring alternative energy storage methods that offer enhanced safety, environmental friendliness, and unique properties. Some of these promising alternatives include:
- Fuel Cells: Fuel cells, particularly hydrogen-oxygen cells, utilize the oxidation of hydrogen and reduction of oxygen to generate electric current, producing water as a byproduct. While they offer zero emissions, the current cost and hydrogen production challenges limit their widespread use. Efficient and clean hydrogen production is a key challenge in fuel cell technology.
- Graphene Supercapacitors: Supercapacitors store energy electrostatically and can be charged and discharged rapidly compared to chemical batteries. Although their energy density is currently lower than batteries, graphene-based supercapacitors hold the potential to overcome this limitation and offer higher energy density in the future.
- Flow Batteries: Flow batteries store energy in chemical electrolytes, with redox reactions being a common mechanism. They are known for their long service life and scalability. However, they suffer from lower energy density and slower charge/discharge rates compared to some other technologies.
- Aluminum-Graphite Batteries: These batteries are safe and lightweight, enabling rapid charging. Currently, their voltage output is insufficient for powering vehicles or phones, but ongoing research aims to enhance this aspect.
- Bioelectrochemical Batteries: Bioelectrochemical batteries employ anaerobic bacteria that process acetate using redox reactions, leading to electric current generation. While this technology is in its early stages with limited operating cycles, future developments may lead to batteries with nearly infinite lifespans through bacterial multiplication.
- These emerging energy storage technologies offer diverse possibilities for meeting specific energy storage needs, whether through improved performance, cost-effectiveness, or sustainability, and they represent exciting avenues for the future of energy storage solutions.
The future of lithium-ion batteries
Despite the emergence of various battery technologies, it’s unlikely that any of them will surpass lithium-ion batteries in the near future. While there is ongoing potential for chemical and technological advancements to achieve higher energy density and longer battery life with lithium-ion batteries, there are concerns about the rapidly diminishing global resources of the raw materials required for their production. Consequently, the development of alternative technologies is becoming increasingly necessary to address both resource sustainability and the growing demand for energy storage solutions.