Flow batteries

Flow batteries
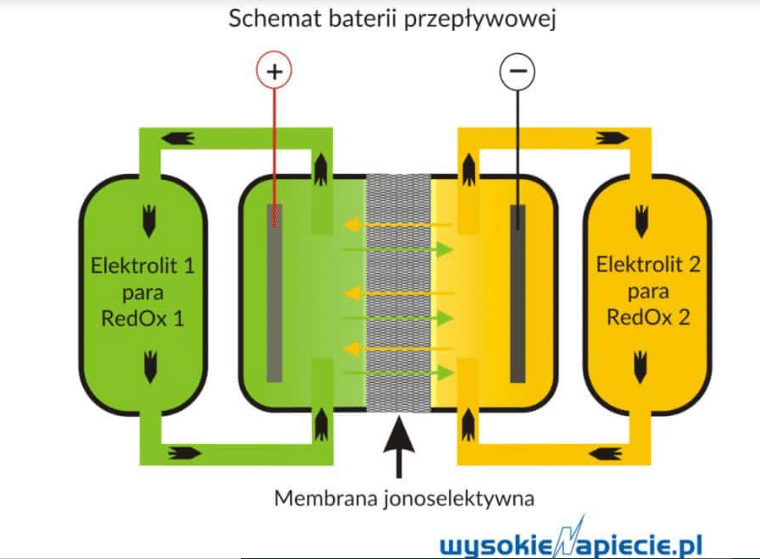
Flow batteries, a popular energy storage option, work by storing energy in two liquid chemical solutions. These solutions flow through an electrochemical cell on different sides of a special membrane. This membrane keeps the solutions separate while allowing ions to move between them, which converts chemical energy into usable electrical energy.
Flow batteries, a popular energy storage option, work by storing energy in two liquid chemical solutions. These solutions flow through an electrochemical cell on different sides of a special membrane. This membrane keeps the solutions separate while allowing ions to move between them, which converts chemical energy into usable electrical energy.
source: High Voltage, CC BY 4.0
Flow batteries are gaining popularity because their components are cost-effective, and they can be scaled up as needed. They offer high power density from cells and can store energy for extended periods. The power capacity depends on the cell’s size and shape, while the amount of stored energy is determined by the tank size. This flexibility allows for custom energy storage solutions. Flow batteries can serve a wide range of applications, from small-scale setups to large-scale projects. Their durability means they can last up to 30 years. As a result, flow batteries might eventually surpass the commonly used lithium-ion batteries in various applications.
Battery redox
Redox flow batteries, particularly vanadium (V/V) batteries, are currently the most popular type of flow batteries. These batteries rely on reduction and oxidation reactions, which involve one reactant losing electrons (reduction) while the other gains them (oxidation). This chemical reaction generates electron movement, which flows through an external circuit to power electrical devices. What’s noteworthy is that the redox reaction is reversible after discharge, enabling the reactants’ oxidation state to be reset for reuse. Redox batteries can be rapidly “recharged” by replacing the used electrolytes in the tanks with fresh ones.
In the case of vanadium flow batteries, one half-cell changes its oxidation state using vanadium oxide ions (VO2+ and VO2+), while the other involves vanadium ions (V3+ and V2+). These batteries are valued for their large capacity, making them excellent for storing energy from renewable sources, and their energy density, making them suitable for Uninterruptible Power Supply (UPS) systems. Additionally, their low self-discharge enables long-term energy storage applications. Vanadium batteries can be deployed on a broader scale for regulating grid frequency and smoothing load fluctuations.
It’s important to note that various other electrolyte pairs, such as bromine and sulfur (Br/S), iron and chromium (Fe/Cr), or zinc and bromine (Zn/Br), can be used in redox batteries as well.
Hybrid flow faucets
Hybrid flow batteries merge the attributes of traditional batteries and flow batteries. In these batteries, one of the active substances is stored in the electrochemical cell, while the other is stored as a liquid electrolyte in an external tank. The duration of energy storage in such flow batteries depends on both the electrolyte volume and the electrode surface and can typically last from 6 to 12 hours.
Hybrid flow battery developments include Zn-Ce (zinc-cerium) and Zn-Br2 (zinc-bromine) batteries. In the discharge process, zinc undergoes oxidation (loses electrons), and during charging, it experiences reduction (gains electrons). Zinc-bromine batteries, in particular, show promise as an alternative to widely used lithium-ion batteries due to their ability to discharge fully without harming the cell.
Other types of flow batteries
Flow batteries encompass membraneless batteries in which electrolytes aren’t separated. By ensuring that the flow of electrolytes maintains laminarity as they pass through the channel where the reactions occur, the mixing of substances is restricted. This setup permits the use of substances whose combination would rapidly deteriorate a membrane, for instance, bromine and hydrogen. Another variety includes batteries that utilize networks of nanoparticles, enabling electric current to travel through liquid substances, which can result in higher energy yield. Certain batteries incorporate not only liquid substances but also solids, specifically semi-solids, targeted for redox reactions. These diverse approaches offer unique characteristics and applications within the realm of flow batteries.
Directions of development of flow batteries
Currently, the most advanced flow battery is the vanadium battery, known for the stability of its ions. It can withstand numerous charging and discharging cycles without unwanted reactions. However, it does have a drawback of lower energy density compared to lithium-ion batteries. Furthermore, vanadium is a costly material, which underscores the importance of exploring alternative redox batteries.
Efforts to enhance the energy density of flow batteries include research into organic electrolytes like viologen. Some researchers are also working on an intermediate battery concept, bridging the gap between flow batteries and lithium-ion batteries. These batteries feature external tanks for energy storage, similar to conventional flow batteries, but instead of liquid electrolytes, they contain solid cathode material (like lithium iron phosphate, or LFP) and titanium dioxide as the anode material. Ions and electrons are transported between these materials using liquid mediators. Additionally, semi-solid batteries are in development.
There are continuous advancements in the materials used for separation membranes. Scientists have modified existing materials like Nafion by combining them with other polymers, allowing for improved penetration of lithium ions through the membrane.
Flow batteries represent a promising future technology. Ongoing research and development efforts are continually refining existing battery types and introducing new and innovative approaches in the realm of energy storage.